Mpox: Stumbling Over the Same Stone Again
2024
Smallpox was a devastating disease that plagued humanity for millennia, causing millions of deaths over the more than 3,000 years it coexisted with people. However, it is an exception in humanity’s battle against infectious diseases, as it is the only one to have been successfully eradicated. In 1796, Jenner created the world’s first vaccine, designed to protect against smallpox. In 1967, World Health Organization (WHO) launched an intensive strategy focused on immunisation and case monitoring, leading to the disease’s eradication by 1980. As a result, there was no longer a need to continue vaccinating the population, and some countries, such as the United States, had even ceased vaccinations by 1972, years prior to the official declaration of eradication.
Mpox (formerly known as monkeypox), which has captured attention in recent years, is not a new disease. It was first identified in 1958 in Denmark, in colonies of monkeys bred for research purposes. The first human case was reported in 1970 in a nine-month-old boy from a region of the Democratic Republic of Congo (DRC), where smallpox had been eradicated two years earlier. Mpox is a viral zoonosis, meaning it is transmitted to humans from animals, with symptoms resembling those seen in smallpox patients. However, it is generally much less severe. While smallpox had a case fatality rate of up to 30%, mpox typically results in much lower (but no less concerning) fatality rates.
Mpox is usually self-limiting, meaning it typically resolves on its own within 2 to 4 weeks. Its clinical manifestations can vary widely, usually appearing 5 to 21 days after exposure. The most characteristic symptom is the appearance of rashes, which may be few or numerous. These start as blotches and progress to fluid-filled vesicles that may be itchy or painful, with a risk of serious infection if left untreated. Over time, the vesicles dry out, crust over and eventually fall off. Early symptoms may also include non-specific signs such as high fever, headache and muscle aches, which can complicate diagnosis since they resemble those of many other diseases. Complications of mpox, in addition to skin infections like cellulitis, may include secondary infections such as bronchopneumonia, sepsis, encephalitis, or corneal infections that can lead to vision loss. PCR testing provides the most accurate diagnosis, but access to this test is limited in rural areas of Africa. In Africa, mpox virus has been found in a variety of animals, including squirrels, rats, dormice and different monkey species, which increases its potential for spread. Animal-to-human (zoonotic) transmission occurs through direct contact with the blood, body fluids, or skin and mucosal lesions of infected animals, as well as from person to person. Person-to-person transmission typically results from close contact with respiratory secretions, skin lesions, or recently contaminated objects. Respiratory transmission via droplets usually requires prolonged face-to-face contact, which puts healthcare workers and household members at increased risk. Transmission can also occur through the placenta, potentially leading to congenital mpox, or during close contact at or after birth. Like smallpox, mpox spreads through direct contact, including kissing, touching and sexual intercourse. Those most at risk for severe disease or complications include pregnant women, children and individuals with weakened immune systems. Severe cases are often linked to the extent of exposure to the virus, the patient’s overall health and the presence of any complications.
Mpox is caused by a virus belonging to the Orthopoxvirus genus, which also includes the virus responsible for human smallpox. There are two distinct genetic clades (or variants) of the virus: Clade I, which now has subclades Ia and Ib, is associated with the Congo Basin and tends to be more virulent, while Clade II, which includes subclades IIa and IIb, originates in West Africa and typically causes less severe disease. Vaccines designed to combat a potential resurgence of smallpox appear to offer protection against mpox, though there is no evidence of this in minors.
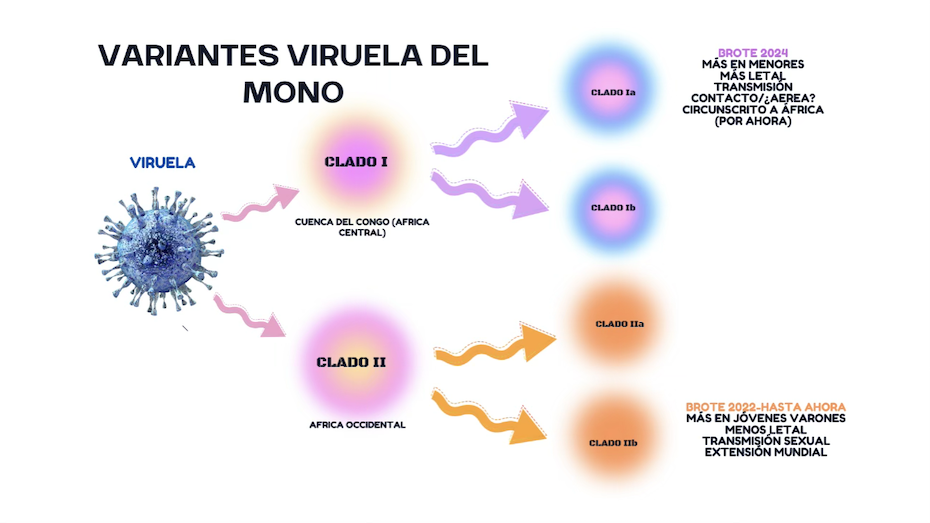
Until 2022, mpox was confined to West and Central Africa, attracting limited international attention. However, in May 2022, in addition to the usual African countries, clusters of mpox cases were detected in several non-endemic countries, including Spain. WHO declared a public health emergency in July 2022, when 75 countries had already been affected. By May 2023, over 87,000 people had been infected worldwide, with 140 deaths reported across 111 affected countries. In response to the steady decline in cases globally, WHO lifted the state of emergency, though it warned that vigilance was still necessary as sporadic cases continued to emerge—insufficient in number to warrant an ongoing international alert. As of 31 August 2024, a total of 106,310 cases and 234 deaths had been confirmed in 123 countries. Cases linked to this outbreak, caused by a Clade IIb strain with specific characteristics, continue to emerge. The most frequent mode of transmission has been sexual, particularly among young or middle-aged men who have sex with men, with an exceptionally low case fatality rate of around 0.5%.
However, the situation changed dramatically in 2023. DRC began experiencing a sharp rise in cases of Clade I. This spread to neighbouring countries, prompting WHO Director-General to declare a Public Health Emergency of International Concern (PHEIC) for the new outbreak of mpox on 14 August 2024. Initially, this outbreak was linked to the Ib clade, with transmission largely occurring through sexual contact. In April 2024, a new subvariant, Clade Ia, was identified for the first time in South Kivu, DRC, exhibiting distinct characteristics—possibly a mutation due to inadequate outbreak control in the region. Much remains unknown about this subvariant, but it is known to affect children more severely, to be more deadly and to have a higher likelihood of airborne transmission. It is associated with a case fatality rate of 3%. Children under 15 years of age now account for over 65% of confirmed cases and more than 80% of deaths. While no cases have been reported outside the region, the risk of spread is significant and health authorities worldwide have been alerted. The Centre for Disease Control and Prevention (CDC) in Africa reported 6,806 confirmed cases of mpox, 34,297 suspected cases and 866 deaths from January to September 2024, with a case fatality rate of 2.52%. These figures likely underrepresent the true number of cases, largely due to diagnostic challenges in isolated areas.
This situation has led to a consensus in the international community that all available tools must be used to tackle the outbreak, including vaccines—despite the lack of evidence that they would be effective in protecting children. On 13 September 2024, WHO prequalified the first mpox vaccine (Bavarian Nordic’s MVA) for use in this outbreak, claiming that “vaccination will play a key role in combating the outbreak”. This pre-qualification came nearly a month after Dr. Kaseya, Director of CDC Africa, called on 17 August for 10 million doses of vaccine to be sent to Africa in 2025, with an additional two million doses for the remainder of 2024.
Dr. Kaseya had previously remarked that Africa did not receive the urgent support it needed during the 2022 outbreak. This was due to a lack of international interest once global cases had declined, even though the situation continued to worsen in Africa. He urged the international community to refocus its efforts on supporting African countries in this new outbreak. In addition to this lack of interest from wealthier nations, several countries hoarded vaccines during the previous outbreak, a response we condemned in our 2022 report. This type of behaviour is not the right approach to tackling any global health crisis, but we seem to have learned little.
So far, the international community has pledged to deliver 3.6 million vaccines to DR Congo, the hardest-hit country, but this is only 36% of the amount requested by the African CDC. Of these, 620,000 doses are from the MVA-BN vaccine donated by the European Commission, the United States and several European countries, including Spain. The remaining 3 million doses come from Japan, which donated its LC16 vaccine. While this vaccine has been well received by African authorities, it has not yet been approved by WHO, and there is limited evidence of its effectiveness in epidemic outbreaks, unlike MVA-BN. The issue is not simply getting vaccines to the affected areas. To effectively manage these international health crises, we need to completely rethink our approach to global health.
The first step should be a stronger commitment from the international community to global health, moving beyond local interests and selfishness that either result in lack of care or vaccine hoarding, which delays essential responses on the ground, proving ineffective even for wealthier countries. A well-structured Pandemic Treaty could help improve this situation, strengthening the WHO’s role as the public guarantor of global health and empowering it to take decisive actions. There is also a need for more agile political decision-making to support technical decisions so that action is taken promptly once an outbreak is identified. A solid Pandemic Treaty could facilitate this process.
Countries that are often the first to be affected by outbreaks have fragile health systems that need strengthening. A successful vaccination campaign requires at least adequate transport and storage management, including a reliable cold chain, sufficient trained staff and an informed population. During the initial emergency of this outbreak, vaccines did not arrive immediately in the most affected areas. Furthermore, it is critical to strengthen diagnostic capacity in order to map the evolution of the disease accurately, as well as to improve therapeutic capabilities, which are often limited in rural areas.
Lastly, improving population participation in solutions is vital, not only through access to accurate information but also through more direct involvement in implementing local solutions that can be tailored to specific areas. While this is frequently mentioned in reports, it remains an unrealised goal. We need much more operational tools to address problems like this new mpox outbreak. But we must also reflect on the social, political and economic structures that underpin our societies, structures that not only promote inequity and hinder the search for global and equitable solutions but also allow us to repeat mistakes in short periods, as seen in the management of outbreaks that could potentially threaten humanity.
We must recognise that if this outbreak reaches Western countries and affects children there, the response would not only be more intense, but the interest in controlling the outbreak at its source would also be much greater. The bubble we lived in before COVID-19 in high-income countries burst, and it revealed the necessity for rapid responses to global threats. Yet, it seems we have not learned enough to effectively handle new epidemic outbreaks, whether mpox or any other that may emerge in the future.
In summary, the overall snapshot of global health improvement is not optimistic. The strategies exist, and the problems are well understood, but there is a lack of political will and genuine commitment to achieving the progress we aim for in the next six years. However, there has been considerable progress, allowing us to affirm that even if the proposed targets are not fully reached, the effort to get as close as possible is worthwhile. It will prevent many avoidable deaths and suffering and may guide us in continuing our work beyond 2030.